
Polymers
Our drug delivery excipients & medical material
Bioabsorbable and biocompatible PLA and PLGA polymers
We can provide our customers with small or bulk quantities for screening of PLA/PLGA ± PEG polymers (up to 50 references available) to improve formulation at early stages of development. These polymers are manufactured according to GMP standards.
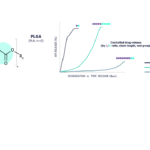
Drug-release can be controlled by the Lactic acid/Glycolic acid monomer ratio, chain length or end groups. Typically longer chain length and higher lactic acid monomer ratio are associated with slower degradation / drug release. These adjustable properties make our unique grades and tailor-made grades.
Learn more about SEQENS Expansorb® PLGA grades range: 20+ years of expertise in their development and cGMP manufacturing
PEKK polymers
These polymers are widely used in orthopedics, as well as neurological, dental, spinal and cardiovascular implants but also in PEKK-based 3D-printed bones. Catalog on demand
Seqens can also develop other GMP medical grade polymers such as hydroxybutyrate, (poly)ester, acrylate and methacrylate copolymers for our customers.
Custom polymers for pharmaceutical applications
Polymeric APIs or custom functionalized polymers for drug delivery.
For custom polymers development and manufacturing, Seqens has a strong experience and offers to its customers:
- Various Chemical Reactions Expertise
- Flexibility in manufacturing proposals
- Repeatability and robustness of performances